Most materials have a rough, matte surface texture, which is what makes the luster of metals so unique and special. Not only do metals have a luster, but they can also be polished to have a mirror finish, in fact, mirrors reflect light by utilizing the reflectiveness of a smooth metal surface.
But why are metals reflective while most materials are not?
When light encounters a rough surface — which we perceive as matte — the light is scattered in many different directions, therefore the rough surface appears to give off a diffuse glow rather than a reflected image.
When light encounters a nearly perfectly smooth surface, it will most often reflect off the surface with the same angle at which it encountered the surface. While this does produce a reflected image, the image is usually somewhat foggy, blurry, or diffuse.
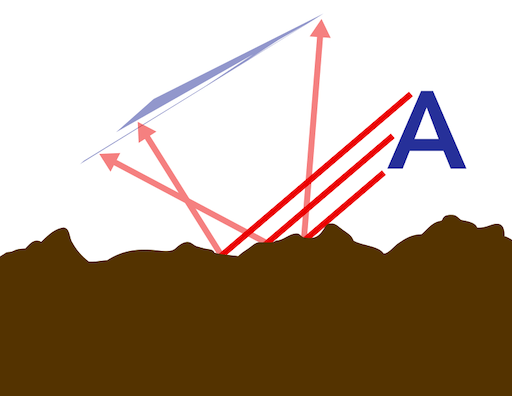
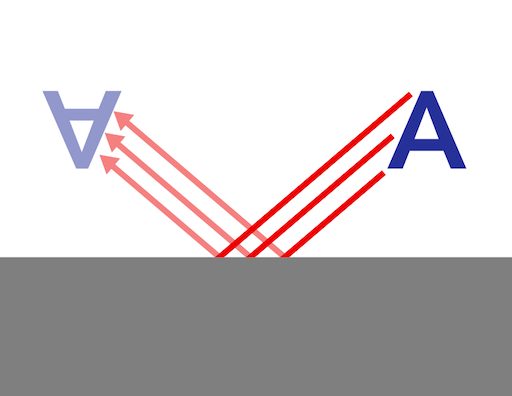
If that smooth surface is made of metal, however, the light is normally reflected almost perfectly, which causes the metal to have a mirror finish because the light maintains its angle of incidence —that is merely the scientific way of saying that the light is reflected with the same angle with which it encountered the surface, relative to a line perpendicular to the surface.
Metals typically diffuse light much less than most surfaces, which causes them to appear shinier than most surfaces. This occurs because metals arrange their atoms into a crystal lattice, however, the atoms’ valance electrons delocalize and flow across the surface of the metal in a “sea of electrons.” These free-flowing electrons maintain the light’s angle of incidence when they reemit light that strikes the metal’s surface.
What do you think about shiny surfaces?

